

Our company`s evolution over time

Our company’s history begins in December 1955, with the foundation of the first factory in Romania and South-East Europe producing Penicillin discoverd by Alexander Fleming.
Four years later, the manufacturing plant of Streptomycin comes into operation and the first finished dosage forms – ointments, creams, suppositories – are manufactured.



The technological flows for manufacturing active substances (erythromycin, oxytetracycline, tetracycline, griseofulvin, sinerdol, lysine) are developed.
In this period, Antibiotice becomes the only manufacturer of sterile parenteral (injectable) medicinal products in Romania. The authorization granted in 1977 by the American regulatory body, Food and Drug Administration (FDA) for the manufacturing flow of Streptomycin has opened the doors of the international market.




Antibiotice becomes a joint stock company.
As a result of changes at the macroeconomic level, the Antibiotice`s management reorients the production of medicines.
In our company’s portfolio, finished products gain the first position while active substances manufactured in this period are used for developing new pharmaceutical dosage forms. In a short period of time, more than 30 pharmaceutical products are introduced into manufacturing, ranking the company from Iași among the top manufacturers of anti-infectives in Romania.




Acțiunile Antibiotice (simbol ATB) sunt tranzacționate la categoria I a Bursei de Valori din București;
Antibiotice devine primul producător de medicamente din România care obține certificatul de bună practică de fabricație (GMP) a fluxului de pulberi pentru medicamente injectabile.




The manufacturing flow of Nystatin obtains the FDA authorization, which allows the export of this active substance in the United States of America. Antibiotice is positioned as the world top manufacturer and Nystatin becomes the most important product for export.




Antibiotice’s economic indicators – profit and turnover - record a significant jump, fruit of investments and management policy; a management by objectives system is implemented, designed to increase at all levels our company’s performance.
A new brand identity is launched in order to communicate visually the modernization of our company and the structural changes occurred in the last years.



Our company inaugurates the Center for Medicine Evaluation (CEM), a one million EUR investment. CEM is a clinical research unit conducting non-therapeutic clinical studies (phase I and bioequivalence studies). The Center is Good Laboratory Practice (GLP) certified and authorized by the Ministry of Health.


Antibiotice obtains the recognition for implementing the Integrated Management System (quality, environment, occupational health and safety) according to the requirements of the EN ISO 9001: 2008, EN ISO 14001: 2004 and OHSAS 18001: 2007 standards, being the first Romanian pharmaceutical company with this performance.
Our company completes the investment in an ecological waste incineration plant which, together with the modern wastewater treatment plant, significantly reduce the impact of the company's activities on the environment.


Antibiotice orients itself towards the international development.
Our company obtains the FDA authorization to deliver injectable medicines in the United Sates of America; the number of exported medicinal products increases, as well as the number of international partnerships.
The National Association of Romanian Exporters and Importers (ANEIR) designates Antibiotice as the most dynamic export company in the field of medicines.


Antibiotice is FDA authorized for the manufacturing flow of penicillins in the form of injectable powders and delivers the first finished medicinal products in the USA, a market in which our company was already present with the active substance Nystatin.
Our company starts the ”Summer School a+”, a project intended for the professional development of its own employees, and for attracting future specialists in the fields of research, quality control and production of medicines.
Antibiotice continues its investments in the manufacturing technology (an increased production capacity of oral solid dosage forms - tablets ) and in research, building a modern Research-Development Center meant to increase the pace of renewing the products in the portfolio.
Compania lansează pe piață primele medicamente destinate afecțiunilor sistemului Nervos Central.



Following the FDA 's reapproval of the manufacturing flows of Nystatin and sterile injectable powders (2013) as well as an increased competitiveness, Antibiotice gains the first position in the world production of the active substance Nystatin and records the first export of Nafcillin in the American market.
Our company extends its international presence by opening a representative office in the Republic of Moldova and an office in Serbia.
Antibiotice își extinde portofoliul tradițional de antiinfecțioase, asimilând primele carbapeneme, își diversifică portofoliul de produse dermatologice, cel destinat sistemului nervos central, dar și oferta de produse pentru profilaxie și creșterii calității vieții (suplimente alimentare și medicamente fără prescripție).
Antibiotice becomes the first WHO pre-qualified company in Europe for the range of anti-tuberculosis medicines.




US FDA recertifies the sterile finished injectables and active substance Nystatin.
Our company wins two gold medals and the Prize at EUROINVENT 2015, the Medicine Section, the largest exhibition of inventions and projects in the South-Eastern Europe.
On December 11, 2015, Antibiotice celebrates 60 years of Romanian continuity and performance.




Antibiotice opens a new international office in Hanoi, Vietnam.
Our company starts the „Perform a+” project which focuses on attracting specialized staff and new collaborators in the research & development field. The project is dedicated to the young students and residents of the Faculty of Pharmacy within the Gr. T. Popa University of Medicine and Pharmacy Iași.



Antibiotice Iași imposes worldwide the quality standard for Nystatin, our product becoming a USP reference standard.




Our third international representative office is opened in Kiev, Ukraine.
Antibiotice starts the serialization of its medicines, making the first delivery of serialized products in the US.
Antibiotice is the first pharmaceutical company joining the Romanian Investors Relations Association (ARIR).
Laboratory Management System (LMS) is implemented.
The production of Moldamin, the new formula of benzathine benzylpenicillin is resumed.



1955-1959
1960-1977
1990-1997
1997-1999
2000-2004
2005
2006
2007
2009
2010-2011
2012-2014
2015
2016
2017-2018
2019
Testimonials

„I congratulate Antibiotice on the anniversary, primarily for its efforts to support the Romanian health system during its 65 years...”
continue readingDr. Diana Loreta Păun, Presidential Advisor, Department of Public Health

„We are honored to send you a message of support and congratulation on the occasion of the 65th anniversary of Antibiotice Iași, the most famous Romanian brand of medicinal products....”
continue readingCristina Racoceanu, President, the National Agency for Medicines and Medical Devices of Romania

„On behalf of the College of Pharmacists of Romania, it is my honor to extend the sincerest congratulations to the management team and employees of Antibiotice Iasi, on the occasion of the celebration of 65 years since foundation...”
continue readingProf. Dr. Dumitru Lupuliasa, President of the College of Pharmacists of Romania

„I wish to you who are part of the Antibiotice family many happy years!
Since childhood I was one of the beneficiaries of the treatment with Moldamin...”
Dr. Valeria Herdea, Vice President of the Romanian College of Physicians

“Antibiotice is a success story, closely related to the manufactured products, to their evolution from active substances to competitive generic products...”
citește în continuareProfessor Univ. Dr. Adrian Streinu Cercel, Director of the Institute of Infectious Diseases "Matei Bals" Bucharest

“A model of Romanian tradition, continuity and performance, Antibiotice makes every Romanian, every Iasi resident and especially those who constitute the broad soul...”
continue readingProf. Dr. Monica HĂNCIANU, Head of the Council for Doctoral Studies, UMF "Grigore T. Popa", Iași

“All our best wishes and congratulations to an essential pillar of the Romanian industry, with high standards of research and development, with international recognition of the quality and benefits brought...”
continue readingProf.Univ. Dr. Mihaela Cătălina Luca, UMF ”Grigore T. Popa” Iași, Spital Boli Infecțioase “Sf. Parascheva “ Iași

“On the occasion of the 65th anniversary of its existence on the market, I wish the entire Antibiotice Iași team a lot of health and prosperity. A&D Pharma thanks you for the very close partnership developed...”
continue readingCezar Zaharia, CEO A&D Pharma

“Happy birthday, Antibiotice Iași!I am glad to send you all the beautiful thoughts and to congratulate you on the occasion of the beautiful age of 65, of work, innovations and experiments...”
continue readingMaria Cucoreanu , Chief Auditor, TUV Rheinland

“At the anniversary watch of the Antibiotice Company, you receive the best thoughts and good wishes from the team of the Iași Environmental Protection Agency, as an expression of the good collaboration that binds us for 3 decades...”
continue readingGalea Temneanu, Executive Director of the Iași environmental protection Agency

“In the 30 years of my profession I have learned that trust in road partners and respect for each other's contribution is essential...”
continue readingGeorge Daniel Tanasievici, Deputy Chief Inspector of the SSM, territorial labo Inspectorate

“We feel honored to join all those who have come to celebrate, together with you, the 65 years of professionalism and remarkable achievements in the field of medicinal product manufacturing...”
continue readingSteluța Mutescu, General Manager of SC SINPREST SA

“The 65th anniversary of Antibiotice Iași finds it in a process of rebirth, which is a time for launching new projects. Your company has experienced its past 65 years ...”
continue readingDr. Mihai IONESCU, Chief Executive of ANEIR, Co-President of the Export Council of Romania

“Antibiotice Iași is the expression of the fact that WE CAN, in a country that has become too used to saying WE CANNOT. If the companies that Romania had in 1990 had been run as Antibiotice in the last 30 years,..”
continue readingSorin Pâslaru, Editor-in-Chief of Ziarul Financiar

“In order to get an injection, I was immobilized by four clumsy men (one was a militia officer), and my mother said that I, a breath of about seven years at the time, continued to nod and shake and screaming for fear of stinging...”
continue readingMAKE, PDG Press Group BURSA

“Antibiotice Iași is a landmark in Romanian and world medicine. The first factory in Eastern Europe to produce Penicillin discovered by Alexander Fleming, Antibiotice has continuously developed, managing to become today one of the largest Romanian...”
continue readingVirgil Munteanu, Executive Director Evenimentul and Capital Publishing House

“Antibiotice company is a symbol of modernization for Iasi, a testament to the fact that universities and science, which are part of the city's identity structure, find their most useful expression in technology...”
continue readingToni Hrițac, Editor-in-chief of Ziarul de Iași
Anniversary messages
Professor Viorel Scripcariu,
PhD Rector of „Grigore T. Popa” University of Medicine and Pharmacy of Iasi
Luciano CALENTI
Founder & CEO ACIC Pharmaceuticals - Canada
Professor Emeritus Cătălina Arsenescu Georgescu, MD, PhD, FESC, Corresponding Member of the Romanian Academy of Medical Sciences, Honorary Citizen of the City of Iasi
Yu Shen
General Manager International Business, Sinopharm Zhijun - China
Prof. Univ. Dr. Mircea Onofriescu
Manager of the "Cuza-Vodă" Clinical Hospital of Obstetrics and Gynecology
Stefano Fapanni
Senior Sales Director ACS Dobfar, Italy
His Beatitude Patriarch Daniel, Patriarhul The Patriarch of the Romanian Orthodox Church
Paolo Passoni
Sales & Marketing API and Pharma Products, ACS Dobfar, Italy
Iulian Trandafir, General Director Alliance Healthcare Romania
V. Muralidharan (Murali)
President Europe Formulations Business, Aurobindo, India
Alina Barcaru, Group Wholesale Director, Farmexim
Tran Duy Dong , Marketing & Sales Director, Dohapharma - Vietnam
Radu Hanga
President of the Bucharest Stock Exchange
Yelena Fisher
Senior Director Global Alliance Management, Sagent Pharmaceuticals Inc., USA
Daniela Șerban
President of ARIR
Zhuhai United Laboratories – China
Adrian Frantiu
General Manager of TÜV Rheinland Romania
Huang YAN
President Zhijun Sinopharm, China
Daniel Schwartzlose, President, Xellia International - Denmark
Adam Frenc,
Founder & Executive Director Atb Pharma Serbia
Sergio Galošić,
CEO Klimaoprema,
Croatia
Bridge over the Time

Friendship Park a+
The park in front of our company has its roots in 1955, the year in which the factory began its activity. Since then, the curing destiny of Antibiotice included the preoccupation for a healthy lifestyle, care for nature, for environment. After the construction of the factory was completed, the employees planted trees in the area that separated the factory from the road. At first they were thin, limp trees.
Over time, they grew and formed a veritable forest protection curtain. Those fragile saplings have become the trees of today. In 2020, on the occasion of the 65th anniversary of the company, the park was set up and became the Friendship Park a+. With a total surface of 25,000 s.q.m , the park has a wooded area and a lawn with over 1,000 trees and shrubs, divided in three areas:
a recreational space with alleys and benches
a playground for children
a sports field.
Friendship Park a+ is a social responsibility project developed by Antibiotice under the aegis of ”Antibiotice – a friendly brand”, being an arch over time that offers an oasis of relaxation for the community and a playground for children.

The fountain from Antibiotice Iași, the work of the great sculptor Constantin Baraschi
On the occasion of the 65th anniversary of existence, Antibiotice restored the statuary complex located in the square in front of the main entrance, under the close supervision of specialists from the "Moldova" National Museum Complex Iasi. Information about the author of this statue was (re) discovered by the researcher Bobi Apăvăloaei, who has carried out in recent years an analysis of studies and documents on the monuments of Iași.
The work “Monuments from Iași Municipality and County. Bibliographic guide ”, authors: Agrigoroaiei Elidia, Huțanu Georgeta, Kara I, Leibovici Paul, Moscovici Liviu, published in 1969 mentions that the fountain in front of Antibiotice company was created by the sculptor Constantin Baraschi.
Constantin Baraschi (1902-1966) was one of the great sculptors of Romania, with a rich artistic activity, enjoying during his life the recognition of his fellows.', 'antibiotice
After the building of the factory was completed (1953-1955), for embellishing the square at the main entrance, a fountain was placed, a public forum monument. This monument consists of a basin, in the middle of which a pedestal rises, on which the statue of a woman holding on her right shoulder a pitcher is mounted. At the woman`s left leg is a child, both characters looking tenderly at each other.
Although executed during the communist period, this fountain does not carry the symbolic burden specific to the political ideology of the years 1950s-1960s. The statue evokes the theme of motherhood, the sculptor himself showing a deep respect for all mothers.
Constantin Baraschi studied in Bucharest and Paris, learning from renowned professors and in 1937 he was awarded the gold medal for sculpture at the International Exhibition in Paris.
Other monuments that bear his signature are: Kilometre Zero Monument in Bucharest, the Caryatid / Restoration Alley with the Fountain of Modura, bas-reliefs and high reliefs on the Arc de Triomphe.
On the occasion of the 65th anniversary, the Antibiotice company made a gift to the people of Iași and culture lovers, restoring with its own resources the public forum monument, work of the sculptor Constantin Baraschi. The restoration was done according to all scientific rigors. The restoration effort was made by our company in difficult conditions of health crisis, thus demonstrating that the act of culture is possible regardless of the harshness of the times.
a+ today

World leader in the production of Nystatin, a USP reference standard granted by Food and Drug Administration (FDA)
Leader in the segment of sterile injectable powders in Romania
Leader in the segment of topical products (creams, ointments, gels) in Romania
Leader in the segment of generic anti-infectives in Romania

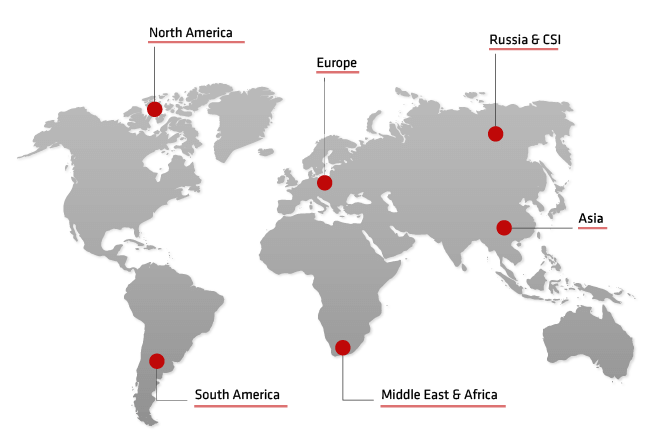
International presence in more than 120 countries with 95 finished products and the active substance Nystatin
International representative offices in Hanoi (Vietnam), Chișinău (Republic of Moldova), Kiev (Ukraine) and Novisad (Serbia)
The export accounts for 50% of the company`s turnover

Sustainable 150-product portfolio covering 12 therapeutic areas
Our company’s products are present in all the hospitals and in over 8,000 pharmacies in Romania
58 medicinal products from the Antibiotice portfolio are considered essential, according to the World Health Organization (WHO) classification
Pre-qualified WHO manufacturer of the full range of essential anti-tuberculosis medicines

Internationally recognized certifications and authorizations (EU-GMP, US-FDA, CoS )
Our company has one of the most modern Clinical Studies Centers in Romania
The most complex manufacturing structure in Romania, with 8 distinct manufacturing flows

A constant presence of over 22 years on the capital market
Since April 1997, Antibiotice shares are listed on the Bucharest Stock Exchange, Premium category

Stimulant work environment for 1,415 employees
Three generations of people with a diversity of professions

For 20 years in a row, our company has been granting the ”Science and Soul ”scholarships dedicated to the rural pupils
For over 10 years, Antibiotice has been organizing the ”Donate blood! Put soul for a life!” campaign in which its employees volunteer to help
10 orchards planted through the program ”The orchard at school


























Antibiotice’s presence in international markets
Anniversary videoconference